Making trials faster & more successful
Research Grid (R.grid) are experts in AI-native automation for clinical trials. Our mission is to make trials admin-free, accelerating access to life-saving medicine. We have safely engineered two industry-leading, fully traceable AI products that together form the only end-to-end solution to automate manual back-office workflows from pre-trial planning to trial close.

Supporting world-leading organizations
The experts in automating clinical trials
Research Grid leverages our AI-native automation engine to safely deliver industry-leading solutions that eliminate trial bottlenecks and automate manual processes. Its unique architecture creates the only end-to-end AI platform engineered specifically for clinical trials, providing full traceability from pre-trial planning to trial close.
Faster
Our smart software cuts months off your trial timeline by connecting every trial stage in one place and automating complex manual workflows using AI
More successful
We make patient recruitment and engagement easy while drastically reducing administrative errors to give every trial the best chance of success

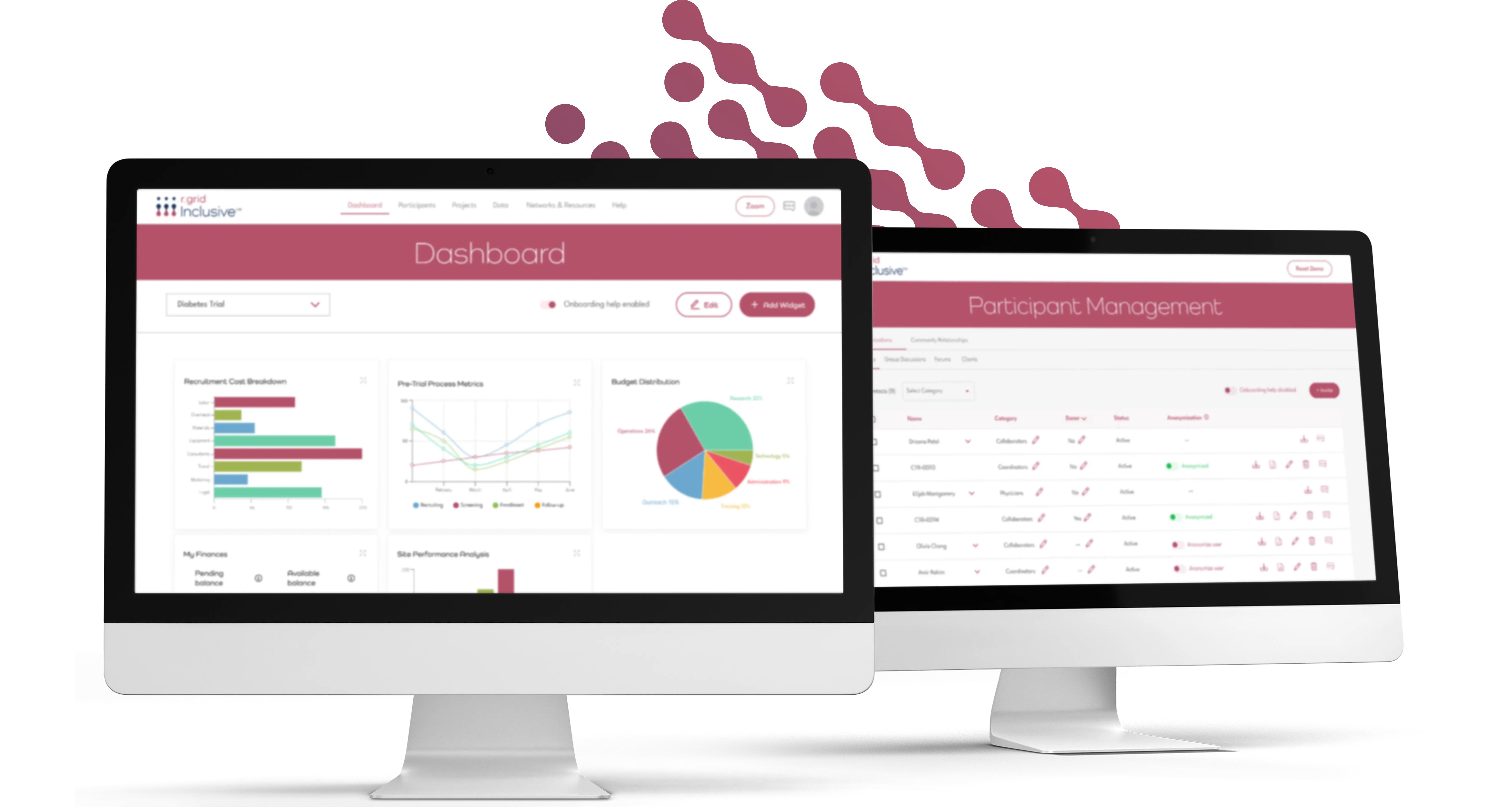
Inclusive
Inclusive is an intelligent patient and community engagement, sourcing, and management platform built for early trial preparation. It’s the first all-in-one solution of its kind, providing access to the world’s largest network of patient communities, with more than 98,000 communities and over 400 million diverse patients globally. Inclusive is powered by AI, making it faster and easier to identify, engage, and manage stakeholders and participants. This reduces delays, strengthens study design, and improves enrolment and retention, preparing trials for success from the very start.

TrialEngine
TrialEngine is a smart clinical trial automation system (CTAS) offering a full suite of eClinical solutions to safely accelerate administration, patient tracking, data management, and reporting workflows during and after trials. It leverages custom AI algorithms and process automation to manage and complete tasks such as protocol development, study documentation and data extraction in a matter of minutes. This improves data quality, unifies workflows, and automates compliance driving trial success.

We make patient recruitment and engagement easy while drastically reducing administrative errors to give every trial the best chance of success.
Global Results

98.5% enrolment rate
across all trials
73% of enrolled patients
belong to ethnic minorities
99.25% patient retention
through automation and engagement
98.5% enrolment rate across all trials
73% of enrolled patients belong to ethnic minorities
99.25% patient retention through automation and engagement
98.5% enrolment rate across all trials
73% of enrolled patients belong to ethnic minorities
99.25% patient retention through automation and engagement
The Future of Clinical Trials Isn't Just AI-Assisted, It's AI-Native
Research Grid has built the first automation engine designed for the clinical trial back-office end-to-end. We ensure our clinical trial partners have the technology and systems that are scalable, efficient, and equitable. We do this by automating bottlenecks for participants, data, and teams. The result unlocks faster study enrollment, increased patient retention, cleaner data, and quality insights that improve research.
What Our Partners Say
I’m loving all of the community networks that Inclusive has to offer in the database! The ease of sending mass emails is a huge plus!

A.F
Senior Manager of Patient Service, EmVenio
We have found a partner in Research Grid’s Inclusive platform that simply understands us. There is an intimate level of knowledge that can only be derived from experience.

N.B
Director Global Patient Engagement
TrialEngine’s document automation is truly a game changer and significantly increases our agility.

Dr N.S.
CEO, Pharmaceutical simulation supplier, former site Principal Investigator
Working with the Inclusive team was fun, efficient and effective. They are doing everything in a short time and making it simple. Inclusive itself is an interesting platform that allows us in Indonesia to connect with other communities and researchers in the world who share the same focus.

S.S.
Indonesian SMA Community-Komunitas Spinal Muscular Atrophy
I love the platform. It actually has the potential to solve real problems in clinical trial operations and patient engagement and retention.

D.W
Vice President, Law Data Enablement Johnson & Johnson Medtech
Inclusive plays a noticeable role in today`s world of globalization. Different community and research groups from all around the world are being brought together on one platform, allowing easier connection and communication.

M.H.
Friedreich Ataxia Lebanese Association (FALA)
This technology has the potential to dramatically improve the efficiency and accuracy of how we perform clinical trials and clearly emphasizes the role of digital technologies in supporting research workflows.

Prof. A.M.
Queen Mary’s William Harvey Research Institute
Partnering with Research Grid to support our patient sourcing has made it easy to quickly connect with and engage patients from a variety of backgrounds across several therapeutic areas.

B. T.
Scientific and Medical Project Manager, Kayentis


























